Cortimax 12 mg Tablets
Therapy Area
Corticosteroids
1.0 Generic name
Deflazacort Tablets 6 mg/12mg/24 mg/30 mg
2.0 Qualitative and quantitative composition
Each uncoated tablet contains
Deflazacort 6 mg/12 mg/24 mg/30 mg.
Excipients q.s.
3.0 Dosage form and strength
Tablets for oral administration.
6 mg/12 mg/24 mg/30 mg
4.0 Clinical particulars
4.1. Therapeutic indication
For asthma, rheumatoid arthritis when glucocorticosteriod therapy is warranted.
4.2.Posology and method of administration
Deflazacort is a glucocorticoid derived from prednisolone and 6mg of deflazacort has approximately the same anti-inflammatory potency as 5mg prednisolone or prednisone. Doses vary widely in different diseases and different patients. In more serious and lifethreatening conditions, high doses of deflazacort may need to be given. When deflazacort is used long term in relatively benign chronic diseases, the maintenance dose should be kept as low as possible. Dosage may need to be increased during periods of stress or in exacerbation of illness. The dosage should be individually titrated according to diagnosis, severity of disease and patient response and tolerance. The lowest dose that will produce an acceptable response should be used.
Adults
For acute disorders, up to 120 mg/day deflazacort may need to be given initially. Maintenance doses in most conditions are within the range 3-18 mg/day. The following regimens are for guidance only
Rheumatoid arthritis: The maintenance dose is usually within the range 3 - 18 mg/day. The smallest effective dose should be used and increased if necessary.
Bronchial asthma: In the treatment of an acute attack, high doses of 48-72 mg/day may be needed depending on severity and gradually reduced once the attack has been controlled. For maintenance in chronic asthma, doses should be titrated to the lowest dose that controls symptoms.
Other conditions: The dose of deflazacort depends on clinical need titrated to the lowest effective dose for maintenance. Starting doses may be estimated on the basis of ratio of 5mg prednisone or prednisolone to 6mg deflazacort.
Hepatic Impairment
In patients with hepatic impairment, blood levels of deflazacort may be increased. Therefore, the dose of deflazacort should be carefully monitored and adjusted to the minimum effective dose.
Renal Impairment
In renally impaired patients, no special precautions other than those usually adopted in patients receiving glucocorticoid therapy are necessary.
Elderly
In elderly patients, no special precautions other than those usually adopted in patients receiving glucocorticoid therapy are necessary. The common adverse effects of systemic corticosteroids may be associated with more serious consequences in old age.
Paediatric Population
There has been limited exposure of children to deflazacort in clinical trials. In children, the indications for glucocorticoids are the same as for adults, but it is important that the lowest effective dosage is used. Alternate day administration may be appropriate.
Doses of deflazacort usually lie in the range 0.25 - 1.5 mg/kg/day. The following ranges provide general guidance:
Juvenile chronic arthritis: The usual maintenance dose is between 0.25 - 1.0 mg/kg/day
Nephrotic syndrome: Initial dose of usually 1.5 mg/kg/day followed by down titration according to clinical need.
Bronchial asthma: On the basis of the potency ratio, the initial dose should be between 0.25 - 1.0 mg/kg deflazacort on alternate days.
Deflazacort withdrawal
In patients who have received more than physiological doses of systemic corticosteroids (approximately 9mg per day or equivalent) for greater than 3 weeks, 3 withdrawal should not be abrupt. How dose reduction should be carried out depends largely on whether the disease is likely to relapse as the dose of systemic corticosteroids is reduced. Clinical assessment of disease activity may be needed during withdrawal. If the disease is unlikely to relapse on withdrawal of systemic corticosteroids but there is uncertainty about HPA suppression, the dose of systemic corticosteroids may be reduced rapidly to physiological doses. Once a daily dose equivalent to 9mg deflazacort is reached, dose reduction should be slower to allow the HPA-axis to recover. Abrupt withdrawal of systemic corticosteroid treatment, which has continued up to 3 weeks is appropriate if it is considered that the disease is unlikely to relapse. Abrupt withdrawal of doses up to 48 mg daily of deflazacort, or equivalent for 3 weeks is unlikely to lead to clinically relevant HPA-axis suppression, in the majority of patients. In the following patient groups, gradual withdrawal of systemic corticosteroid therapy should be considered even after courses lasting 3 weeks or less:
- Patients who have had repeated courses of systemic corticosteroids, particularly if taken for greater than 3 weeks.
- When a short course has been prescribed within one year of cessation of long-term therapy (months or years).
- Patients who may have reasons for adrenocortical insufficiency other than exogenous corticosteroid therapy.
- Patients receiving doses of systemic corticosteroid greater than 48 mg daily of deflazacort (or equivalent),
- Patients repeatedly taking doses in the evening
4.3.Contraindications
Systemic infection unless specific anti-infective therapy is employed. Hypersensitivity to the active substance, deflazacort or any of the excipients. Patients receiving live virus immunisation.
4.4.Special warnings and precautions for use
Patients with rare hereditary problems of galactose intolerance, the Lapp lactose deficiency or glucose-galactose malabsorption should not take this medicine. Undesirable effects may be minimised by using the lowest effective dose for the minimum period, and by administering the daily requirement as a single morning dose or whenever possible as a single morning dose on alternate days. Frequent patient review is required to appropriately titrate the dose against disease activity.
Adrenal suppression
Adrenal cortical atrophy develops during prolonged therapy and may persist for years after stopping treatment. Withdrawal of corticosteroids after prolonged therapy must therefore always be gradual to avoid acute adrenal insufficiency which could be fatal, being tapered off over weeks or months according to the dose and duration of treatment. During prolonged therapy, any intercurrent illness, trauma or surgical procedure will require a temporary increase in dosage; if corticosteroids have been stopped following prolonged therapy, they may need to be temporarily re-introduced. Patients should carry 'Steroid treatment' cards which give clear guidance on the precautions to be taken to minimise risk and which provide details of prescriber, drug, dosage and the duration of treatment.
Anti-inflammatory/immunosuppressive effects and infection
Suppression of the inflammatory response and immune function increases the susceptibility to infections and their severity. The clinical presentation may often be atypical and serious infections such as septicaemia and tuberculosis may be masked and may reach an advanced stage before being recognised. Chickenpox is of particular concern since this normally minor illness may be fatal in immunosuppressed patients. Patients (or parents of children) without a definite history of chicken pox should be advised to avoid close personal contact with chickenpox or herpes zoster and, if exposed, they should seek urgent medical attention. Passive immunisation with varicella zoster immunoglobulin (VZIG) is needed by exposed non-immune patients who are receiving systemic corticosteroids or who have used them within the previous 3 months; this should be given within 10 days of exposure to chickenpox. If a diagnosis of chickenpox is confirmed, the illness warrants specialist care and urgent treatment. Corticosteroids should not be stopped and the dose may need to be increased. Patients should be advised to take particular care to avoid exposure to measles and to seek immediate medical advice if exposure occurs. Prophylaxis with intramuscular normal immunoglobulin may be needed. Live vaccines should not be given to individuals with impaired responsiveness. The antibody response to other vaccines may be diminished.
Visual disturbance
Visual disturbance may be reported with systemic and topical corticosteroid use. If a patient presents with symptoms such as blurred vision or other visual disturbances, the patient should be considered for referral to an ophthalmologist for evaluation of possible causes which may include cataract, glaucoma or rare diseases such as central serous chorioretinopathy (CSCR) which have been reported after use of systemic and topical corticosteroids. Prolonged use of glucocorticoids may produce posterior subcapsular cataracts, glaucoma with possible damage to the optic nerves and may enhance the establishment of secondary ocular infections due to fungi or viruses. Use in active tuberculosis should be restricted to those cases of fulminating and disseminated tuberculosis in which deflazacort is used for management with appropriate antituberculosis regimen. If glucocorticoids are indicated in patients with latent tuberculosis or tuberculin reactivity, close observation is necessary as reactivation of the disease may occur. During prolonged glucocorticoid therapy, these patients should receive chemoprophylaxis. Tendonitis and tendon rupture are known class effect of glucocorticoids. The risk of such reactions may be increased by co-administration of quinolones. Pheochromocytoma crisis, which can be fatal, has been reported after administration of systemic corticosteroids. Corticosteroids should only be administered to patients with suspected or identified pheochromocytoma after an appropriate risk/benefit evaluation.
Special precautions
The following clinical conditions require special caution and frequent patient monitoring is necessary: -
- Cardiac disease or congestive heart failure (except in the presence of active rheumatic carditis), hypertension, thromboembolic disorders. Glucocorticoids can cause salt and water retention and increased excretion of potassium. Dietary salt restriction and potassium supplementation may be necessary.
- Gastritis or oesophagitis, diverticulitis, ulcerative colitis if there is probability of impending perforation, abscess or pyogenic infections, fresh intestinal anastomosis, active or latent peptic ulcer.
- Diabetes mellitus or a family history, osteoporosis, myasthenia gravis, renal insufficiency.
- Emotional instability or psychotic tendency, epilepsy.
- Previous corticosteroid-induced myopathy.
- Liver failure.
- Hypothyroidism and cirrhosis, which may increase glucocorticoid effect.
- Ocular herpes simplex because of possible corneal perforation.
Patients and/or carers should be warned that potentially severe psychiatric adverse reactions may occur with systemic steroids. Symptoms typically emerge within a few days or weeks of starting the treatment. Risks may be higher with high doses/systemic exposure, although dose levels do not allow prediction of the onset, type, severity or duration of reactions. Most reactions recover after either dose reduction or withdrawal, although specific treatment may be necessary. Patients/carers should be encouraged to seek medical advice if worrying psychological symptoms develop, especially if depressed mood or suicidal ideation is suspected. Patients/carers should also be alert to possible psychiatric disturbances that may occur either during or immediately after dose tapering/withdrawal of systemic steroids, although such reactions have been reported infrequently.
Particular care is required when considering the use of systemic corticosteroids in patients with existing or previous history of severe affective disorders in themselves or in their first degree relatives. These would include depressive or manic-depressive illness and previous steroid psychosis. Glucocorticoids are known to cause irregular menstruation and leukocytosis, care should be taken with deflazacort.
Paediatric population
Corticosteroids cause dose-related growth retardation in infancy, childhood and adolescence which may be irreversible. Hypertrophic cardiomyopathy has been reported after systemic administration of glucocorticosteroids in preterm infants. In infants receiving administration of systemic glucocorticosteroids, echocardiograms should be performed to monitor myocardial structure and function.
Use in Elderly
The common adverse effects of systemic corticosteroids may be associated with more serious consequences in old age, especially osteoporosis, hypertension, hypokalaemia, diabetes, susceptibility to infection and thinning of the skin. Close clinical supervision is required to avoid life-threatening reactions. Since complications of glucocorticoid therapy are dependent on dose and duration of therapy, the lowest possible dose must be given and a risk/benefit decision must be made as to whether intermittent therapy should be used.
4.5.Interaction with other medicinal products and other forms of interaction
- The same precautions should be exercised as for other glucocorticoids. Deflazacort is metabolised in the liver. It is recommended to increase the maintenance dose of deflazacort if drugs which are liver enzyme inducers are co-administered, e.g. rifampicin, rifabutin, carbamazepine, phenobarbitone, phenytoin, primidone and aminoglutethimide. For drugs which inhibit liver enzymes, e.g. ketoconazole it may be possible to reduce the maintenance dose of deflazacort.
- In patients taking estrogens, corticosteroid requirements may be reduced.
- The desired effects of hypoglycaemic agents (including insulin), anti-hypertensives and diuretics are antagonised by corticosteroids and the hypokalaemic effects of acetazolamide, loop diuretics, thiazide diuretics, beta 2-agonists, xanthines and carbenoxolone are enhanced.
- The efficacy of coumarin anticoagulants may be enhanced by concurrent corticosteroid therapy and close monitoring of the INR or prothrombin time is required to avoid spontaneous bleeding.
- In patients treated with systemic corticosteroids, use of non-depolarising muscle relaxants can result in prolonged relaxation and acute myopathy. Risk factors for this include prolonged and high dose corticosteroid treatment, and prolonged duration of muscle paralysis. This interaction is more likely following prolonged ventilation (such as in the ITU setting).
- The renal clearance of salicylates is increased by corticosteroids and steroid withdrawal may result in salicylate intoxication.
- As glucocorticoids can suppress the normal responses of the body to attack by micro-organisms, it is important to ensure that any anti-infective therapy is effective and it is recommended to monitor patients closely. Concurrent use of glucocorticoids and oral contraceptives should be closely monitored as plasma levels of glucocorticoids may be increased. This effect may be due to a change in metabolism or binding to serum proteins. Antacids may reduce bioavailability; leave at least 2 hours between administration of deflazacort and antacids. 8
- Co-treatment with CYP3A inhibitors, including cobicistat-containing products, is expected to increase the risk of systemic side-effects. The combination should be avoided unless the benefit outweighs the increased risk of systemic corticosteroid side-effects, in which case patients should be monitored for systemic corticosteroid side-effects.
4.6.Fertility, pregnancy and lactation
Pregnancy
The ability of corticosteroids to cross the placenta varies between individual drugs, however, deflazacort does cross the placenta. Administration of corticosteroids to pregnant animals can cause abnormalities of foetal development including cleft palate, intra-uterine growth retardation and effects on brain growth and development. There is no evidence that corticosteroids result in an increased incidence of congenital abnormalities, such as cleft palate/lip in man. However, when administered for prolonged periods or repeatedly during pregnancy, corticosteroids may increase the risk of intra-uterine growth retardation. Hypoadrenalism may, in theory, occur in the neonate following prenatal exposure to corticosteroids but usually resolves spontaneously following birth and is rarely clinically important. As with all drugs, corticosteroids should only be prescribed when the benefits to the mother and child outweigh the risks. When corticosteroids are essential however, patients with normal pregnancies may be treated as though they were in the non-gravid state.
Breast-feeding
Corticosteroids are excreted in breast milk, although no data are available for deflazacort. Doses of up to 50 mg daily of deflazacort are unlikely to cause systemic effects in the infant. Infants of mothers taking higher doses than this may have a degree of adrenal suppression but the benefits of breast feeding are likely to outweigh any theoretical risk.
Fertility
No data is available on Deflazacort and its effects on fertility.
4.7.Effects on ability to drive and use machines
The effect of corticosteroids on the ability to drive or use machinery has not been systematically evaluated. Vertigo is a possible undesirable effect after treatment with deflazacort. If affected, patients should not drive or operate machinery.
4.8.Undesirable effects
The incidence of predictable undesirable effects, including hypothalamic-pituitaryadrenal suppression correlates with the relative potency of the drug, dosage; timing of administration and the duration of treatment. The following CIOMS frequency rating is used: Very common (≥1/10); common (≥1/100 to <1/10); uncommon (≥1/1000 to <1/100); rare (≥1/10000 to <1/1000); very rare (<1/10000), not known (cannot be estimated from the available data).
Endocrine disorders
Uncommon: Suppression of the hypothalamic-pituitary-adrenal axis, amenorrhoea, Cushingoid facies. Not known: Growth suppression in infancy, childhood and adolescence.
Metabolism and nutrition disorders
Common: Weight gain. Uncommon: impaired carbohydrate tolerance with increased requirement for antidiabetic therapy, sodium and water retention with hypertension, potassium loss and hypokalaemic alkalosis when co-administered with beta 2-agonist and xanthines. Not known: Negative protein and calcium balance, increased appetite.
Infections and Infestations
Uncommon: Increased susceptibility and severity of infections with suppression of clinical symptoms and signs, opportunistic infections, recurrence of dormant tuberculosis. Not known: candidiasis.
Musculoskeletal and connective tissue disorders
Uncommon: Osteoporosis, vertebral and long bone fractures. Rare: Muscle wasting. Not known: avascular osteonecrosis, tendonitis and tendon rupture when coadministered with quinolones, myopathy (acute myopathy may be precipitated by nondepolarising muscle relaxants, negative nitrogen balance.
Reproductive system and breast disorders
Not known: Menstrual irregularity.
Cardiac disorders
Not known: Heart failure, hypertrophic cardiomyopathy in preterm infants.
Nervous system disorders
Uncommon: Headache, vertigo.
Not known: restlessness, Increased intra-cranial pressure with papilloedema in children (pseudotumour cerebri), usually after treatment withdrawal, aggravation of epilepsy.
Psychiatric disorders
A wide range of psychiatric reactions including affective disorders such as: Uncommon: depressed and labile mood. Not known: irritable, euphoric, suicidal thoughts. Psychotic reactions including: Not known: mania, delusions, hallucinations, aggravation of schizophrenia Other reactions including: Uncommon: behavioural disturbances. Not known: anxiety, sleep disturbances, and cognitive dysfunction including confusion and amnesia have been reported. Reactions are common and may occur in both adults and children. In adults, the frequency of severe reactions has been estimated to be 5-6%. Psychological effects have been reported on withdrawal of corticosteroids; the frequency is unknown.
Eye disorders
Not known: Vision blurred, increased intra-ocular pressure, glaucoma, papilloedema, posterior subcapsular cataracts especially in children, chorioretinopathy, corneal or scleral thinning, exacerbation of ophthalmic viral or fungal diseases.
Gastrointestinal disorders
Uncommon: Dyspepsia, peptic ulceration, haemorrhage, nausea. Not known: perforation of peptic ulcer, acute pancreatitis (especially in children), candidiasis.
Skin and subcutaneous tissue disorders
Uncommon: hirsutism, striae, acne.
Rare: bruising.
Not known: Skin atrophy, telangiectasia.
General disorders and administration site conditions
Uncommon: Oedema.
Not known: impaired healing.
Immune system disorders
Uncommon: Hypersensitivity including anaphylaxis has been reported.
Blood and lymphatic system disorders
Not known: Leukocytosis.
Vascular disorders
Not known: Thromboembolism in particular in patients with underlying conditions associated with increased thrombotic tendency, rare incidence of benign intracranial hypertension.
Withdrawal symptoms and signs
Not known: Too rapid a reduction of corticosteroid dosage following prolonged treatment can lead to acute adrenal insufficiency, hypotension and death. A 'withdrawal syndrome' may also occur including fever, myalgia, arthralgia, rhinitis, conjunctivitis, painful itchy skin nodules and loss of weight. This may occur in patients even without evidence of adrenal insufficiency.
Class effect
Pheochromocytoma crisis has been reported with other systemic corticosteroids and is a known class effect.
Reporting of suspected adverse reactions
Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via email to: medico@zuventus.com By reporting side effects, you can help provide more information on the safety of this medicine.
4.9. Overdose
It is unlikely that treatment is needed in cases of acute overdosage. The LD50 for the oral dose is greater than 4000 mg/kg in laboratory animals.
5.0 Pharmacological properties
5.1 Pharmacodynamic properties
Pharmacotherapeutic group: corticosteroids for systemic use; Glucocorticoids. ATC code: H02AB13. Deflazacort is a glucocorticoid. Its anti-inflammatory and immunosuppressive effects are used in treating a variety of diseases and are comparable to other antiinflammatory steroids. Clinical studies have indicated that the average potency ratio of deflazacort to prednisolone is 0.69-0.89.
5.2 Pharmacokinetic properties
Absorption: Orally administered deflazacort appears to be well absorbed.
Distribution: The active metabolite D 21-OH achieves peak plasma concentrations in 1.5 to 2 hours. It is 40% protein-bound and has no affinity for corticosteroid-binding-globulin (transcortin).
Biotransformation: Orally administered deflazacort is immediately converted by plasma esterases to the pharmacologically active metabolite (D 21-OH). Metabolism of D 21-OH is extensive. The metabolite of D 21-OH is deflazacort 6- beta-OH.
Elimination: Its elimination plasma half-life is 1.1 to 1.9 hours. Elimination takes place primarily through the kidneys; 70% of the administered dose is excreted in the urine. The remaining 30% is eliminated in the faeces. Metabolism of D 21-OH is extensive; only 18% of urinary excretion represents D 21-OH. The metabolite of D 21-OH, deflazacort 6-beta-OH, represents one third of the urinary elimination.
6.0 Nonclinical properties
Safety studies have been carried out in the rat, dog, mouse and monkey. The findings are consistent with other glucocorticoids at comparable doses. Teratogenic effects demonstrated in rodents and rabbits are typical of those caused by other glucocorticoids. Deflazacort was not found to be carcinogenic in the mouse, but studies in the rat produced carcinogenic findings consistent with the findings with other glucocorticoids.
7.0 Description
Deflazacort, is an oxaline derivative of Prednisolone.
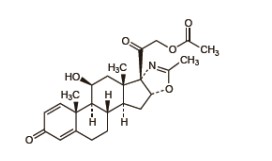
Figure: Structure of Deflazacort
IUPAC Name: (11β,16β)-21-(acetyloxy)-11-hydroxy-2'-methyl-5'H-pregna-1,4- dieno[17,16-d]oxazole-3,20-dione Molecular Formula: C25H31NO6 Molecular Weight: 441.5 g/mol
8.0 Pharmaceutical particulars
1. Incompatibilities Not applicable.
2. Shelf-life
3. Packaging information
4. Storage and handing instructions
- Keep out of reach of children. Keep out of the sight and reach of children. Store in a dry place at a temperature not exceeding 25°C.
9.0 Patient Counselling Information
A patient information leaflet is available for this product.
Administration
Warn patients and/or caregivers to not stop taking Cortimax abruptly or without first checking with their healthcare providers as there may be a need for gradual dose reduction to decrease the risk of adrenal insufficiency.
Cortimax may be taken with or without food. Do not take Cortimax with grapefruit juice.
Tablets
Cortimax Tablets may be taken whole or crushed and taken immediately after mixing with applesauce.
Increased Risk of Infection
Tell patients and/or caregivers to inform their healthcare provider if the patient has had recent or ongoing infections or if they have recently received a vaccine. Medical advice should be sought immediately if the patient develops fever or other signs of infection. Patients and/or caregivers should be made aware that some infections can potentially be severe and fatal. Warn patients who are on corticosteroids to avoid exposure to chickenpox or measles and to alert their healthcare provider immediately if they are exposed.
Alterations in Cardiovascular/Renal Function
Inform patients and/or caregivers that Cortimax can cause an increase in blood pressure and water retention. If this occurs, dietary salt restriction and potassium supplementation may be needed.
Behavioral and Mood Disturbances
Advise patients and/or caregivers about the potential for severe behavioral and mood changes with Cortimax and encourage them to seek medical attention if psychiatric symptoms develop.
Decreases in Bone Mineral Density
Advise patients and/or caregivers about the risk of osteoporosis with prolonged use of Cortimax, which can predispose the patient to vertebral and long bone fractures.
Ophthalmic Effects
Inform patients and/or caregivers that Cortimax may cause cataracts or glaucoma and advise monitoring if corticosteroid therapy is continued for more than 6 weeks.
Vaccination
Advise patients and/or caregivers to bring immunizations up-to-date according to immunization guidelines prior to starting therapy with Cortimax. Live-attenuated or live vaccines should be administered at least 4 to 6 weeks prior to starting Cortimax. Inform patients and/or caregivers that they may receive concurrent vaccinations with use of Cortimax, except for live-attenuated or live vaccines.
Serious Skin Rashes
Instruct patients and/or caregivers to seek medical attention at the first sign of a rash.
Drug Interactions
Certain medications can cause an interaction with Cortimax. Advise patients and/or caregivers to inform their healthcare provider of all the medicines the patient is taking, including over-the-counter medicines (such as insulin, aspirin or other NSAIDS), dietary supplements, and herbal products. Inform patients and/or caregivers that alternate therapy, dosage adjustment, and/or special test(s) may be needed during the treatment.
About leaflet
Important things you need to know about Cortimax
Cortimax is a steroid medicine. This can be prescribed for many different
conditions, including serious illnesses.
You need to take it regularly to get the maximum benefit.
Do not stop taking this medicine without talking to your doctor - you may need to lower the dose gradually.
Cortimax can cause side effects in some people (read section 4 for more information). These include problems such as mood changes (feeling depressed, or ‘high’), or stomach problems, which can happen straight away. If you feel unwell in any way, keep taking your tablets, but see your doctor straight away.
Some side effects only happen after weeks or months. These include weakness of arms and legs, or developing a rounder face (read section 4 for more information).
If you take it for more than 3 weeks, you will be given a blue ‘steroid card’:
always keep it with you and show it to any doctor or nurse treating you.
Keep away from people who have chickenpox, measles or shingles, if you have never had them. They could affect you severely. If you do come into contact with chickenpox or shingles, see your doctor straight away.
Now read the rest of this leaflet. It includes other important information on the safe and effective use of this medicine that might be especially important for you.
Read all of this leaflet carefully before you start taking this medicine
- Keep this leaflet. You may need to read it again.
- If you have any further questions, please ask your doctor or pharmacist.
- This medicine has been prescribed for you. Do not give it to others. It may harm them, even if their symptoms are the same as yours.
- If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
- Your doctor may have given you this medicine before from another company. It may have looked slightly different. However, either brand will have the same effect.
In this leaflet:
- What Cortimax is and what it is used for
- Before you take Cortimax
- How to take Cortimax
- Possible side effects
- How to store Cortimax
- Further information
1. What Cortimax is and what it is used for
The name of your medicine is Cortimax 6mg/12mg/24mg/30mg Tablets (called Cortimax throughout this leaflet). Cortimax is a steroid medicine. Their full name is glucocorticoids.
How Cortimax works
- These corticosteroids occur naturally in the body, and help to maintain health and wellbeing.
- Boosting your body with extra corticosteroid (such as Cortimax) is an effective way to treat various illnesses involving inflammation in the body.
- Cortimax works by reducing this inflammation, which could otherwise go on making your condition worse.
- Cortimax also works by stopping reactions known as autoimmune reactions. These reactions happen when your body’s immune system attacks the body itself and causes damage.
- You must take this medicine regularly to get maximum benefit from it.
Cortimax can be used to:
- Treat inflammation including asthma, arthritis and allergies.
- Treat problems with your skin, kidney, heart, digestive system, eyes or blood.
- Suppress the immune system in transplant operations.
2. Before you take Cortimax
Do not take this medicine and tell your doctor if:
- You are allergic (hypersensitive) to deflazacort or any of the other ingredients in these tablets (see Section 6: Further information). Signs of an allergic reaction include: a rash, swallowing or breathing problems, swelling of your lips, face, throat or tongue.
- You have an infection that affects your whole body (systemic infection), which is not already being treated.
- You are having or have recently had any vaccinations with live viruses (see “vaccinations” below).
Do not take this medicine if any of the above apply to you. If you are not sure, talk to your doctor or pharmacist before taking Cortimax.
Take special care and check with your doctor before you take Cortimax if:
- You have ever had severe depression or manic-depression (bipolar disorder). This includes having had depression before while taking steroid medicines like Cortimax.
- Any of your close family has had these illnesses.
- You have or ever had mental problems such as depression or psychoses. If any of the above applies to you, talk to a doctor before taking Cortimax.
Mental problems while taking Cortimax
Mental health problems can happen while taking steroids like Cortimax (see also section 4 Possible Side Effects).
- These illnesses can be serious.
- Usually they start within a few days or weeks of starting the medicine.
- They are more likely to happen at high doses.
- Most of these problems go away if the dose is lowered or the medicine is stopped. However, if problems do happen, they might need treatment.
Talk to a doctor if you (or someone taking this medicine), show any signs of mental problems. This is particularly important if you are depressed, or might be thinking about suicide. In a few cases, mental problems have happened when doses are being lowered or stopped
Check with your doctor before taking this medicine if:
- You have epilepsy (fits).
- You or anyone in your family has diabetes.
- You have high blood pressure.
- You have kidney, liver or heart problems.
- You have brittle or weak bones called osteoporosis.
- You have an eye disease that causes detachment of your retina and bulging eyes.
- You or anyone in your family has an eye problem called glaucoma.
- You have an underactive thyroid gland.
- You have problems with your digestive system, including your food pipe (oesophagitis), gut (ulcerative colitis, diverticulitis) or stomach (peptic ulcer).
- You have ever had a bad reaction such as muscle weakness to any steroid.
- You have or ever had an infection caused by a virus or fungus. This includes infections such as athlete’s foot, thrush and cold sores (that may also affect the eye).
- You have or ever had ‘tuberculosis’ (TB).
- You have any problems with your blood vessels such as a blood clot.
- You have a pheochromocytoma (a tumour of adrenal gland tissue. The adrenal glands are located above the kidneys.).
Cortimax may cause inflammation of tendons and easy tearing especially when given together with antibiotics such as ciprofloxacin.
Irregular periods in women and blood problems such as leukocytosis (increase in white blood cells count) may also occur.
If any of the above apply to you, your doctor may want to see you more often during your treatment. Contact your doctor if you experience blurred vision or other visual disturbances.
Taking other medicines
Please tell your doctor or pharmacist if you are taking or have recently taken any other medicines. This includes medicines you obtain without a prescription, including herbal medicines. This is because Cortimax and other medicines can affect the way some other medicines work.
Some medicines may increase the effects of Cortimax and your doctor may wish to monitor you carefully if you are taking these medicines (including some medicines for HIV: ritonavir, cobicistat). In particular, check with your doctor if you are taking any of the following medicines. Your doctor may want to change the dose of Cortimax, or the other medicine.
- Painkillers such as aspirin.
- Aminoglutethimide - used for some types of cancer.
- Ketoconazole - used to treat infections
- Water tablets (diuretics) such as spironolactone, triamterene or amiloride.
- Medicines for thinning your blood (such as warfarin).
- Medicines for diabetes.
- Medicines for epilepsy such as phenobarbitone, primidone, phenytoin, carbamazepine, acetazolamide.
- Medicines which contain oestrogens including oral contraceptives.
- Medicines for tuberculosis (TB) such as rifampicin or rifabutin.
- Medicines for high blood pressure.
- Medicines for indigestion and heartburn (antacids). If you are taking an antacid leave at least 2 hours between taking it and Cortimax.
- Medicines for asthma such as salbutamol and theophylline.
Vaccinations
If you have just had any injections or vaccinations, tell your doctor before you take Cortimax. If you are going to have any injections or vaccinations, tell your doctor or nurse you are taking Cortimax. This includes those needed for a foreign holiday. Some vaccines should not be given to patients taking Cortimax. This is because Cortimax can affect the way some vaccines work.
Operations
If you are going to have an operation, tell your doctor or nurse you are taking Cortimax. Muscle relaxants are sometimes used during an operation or in intensive care unit. Cortimax and muscle relaxants can affect one another.
Pregnancy and breast-feeding
Talk to your doctor before taking Cortimax if:
- You are pregnant, plan to get pregnant, or think you may be pregnant.
- You are breast-feeding, or planning to breast-feed.
Driving and using machines
These tablets may make you feel dizzy, feel like everything around you is spinning, or feel disorientated (vertigo). If this happens, do not drive or use any tools or machines. Cortimax and infections Taking Cortimax can mean that you get infections more easily than usual, and these infections can be more serious.
Chickenpox, measles or shingles
If you get chickenpox, measles or shingles while taking Cortimax, you can become seriously ill.
- Keep away from people who have chickenpox, measles or shingles, if you have never had them. They could affect you severely. If you do come into contact with chickenpox, measles or shingles, see your doctor straight away. Your doctor may want to give you a vaccination to help you from getting these infections.
- If you do catch Chickenpox, measles or shingles, tell your doctor straight away. Your doctor will advise you on how to take Cortimax. You may be told to increase the number of tablets that you use.
3. How to take Cortimax
Always take Cortimax exactly as your doctor has told you. The dose will depend on the illness being treated and any other medicines you are taking. You should check with your doctor or pharmacist if you are not sure.
Taking this medicine
- Take this medicine by mouth.
- Swallow your tablets whole with a glass of water.
- It is important to take your medicine at the right times.
Adults
- The usual dose for most conditions, including rheumatoid arthritis is half to three tablets each day.
- The dose for severe asthma may be up to 8 or 12 tablets each day. This dose may be gradually reduced once the asthma attack has been controlled.
- For some problems up to 20 tablets may be needed each day for several days.
- Children
- Cortimax may be given every day or every other day.
- The doctor will work out the dose based on your child’s age and weight.
- Your child will be given the lowest possible dose.
- The usual dose for chronic arthritis is between 0.25 mg and 1 mg of the medicine for each kg of your child’s bodyweight, each day. The usual dose for kidney problems (nephrotic syndrome) is 1.5 mg of the medicine for each kg of your child’s bodyweight, each day. Depending on how well the medicine works for your child, this dose may then be slowly lowered.
- The usual dose for asthma is between 0.25 mg and 1 mg of the medicine for each kg of your child’s bodyweight, every other day.
- In infants, an echocardiogram (ultrasound) should be performed by the doctor to monitor the structure and function of the muscular tissue of the heart.
Elderly
Your doctor may need to check you more carefully for side effects.
If you take more Cortimax than you should
Tell your doctor or go to the nearest hospital casualty department straight away. Remember to take with you any tablets that are left and the pack. This is so the doctor knows what you have taken.
If you forget to take Cortimax
If you forget to take a dose take it as soon as you remember, unless it is time for your next dose. Do not take a double dose to make up for a forgotten dose.
Stopping treatment
- You need to take Cortimax regularly to get the maximum benefit.
- Do not stop taking this medicine without talking to your doctor – you may need to lower the dose gradually.
- Stopping the treatment suddenly can sometimes cause problems such as a high temperature, a runny nose, sore, red, sticky eyes, aching muscles and joints, itchy skin and weight loss. Also, sickness (vomiting), headaches and drowsiness – this is more likely to happen in children.
You may also notice the following symptoms if you stop treatment with Cortimax.
If this happens, tell a doctor straight away as these could be signs of a serious illness:
- Sudden, severe pain in the back, stomach and legs.
- Being sick (vomiting) and diarrhoea.
- Feeling faint or dizzy, this could be a sign of low blood pressure.
4. Possible side effects
Like all medicines, Cortimax can cause side effects, although not everybody gets them.
Stop taking your medicine and see a doctor or go to a hospital straight away if:
Uncommon (affects 1 to 10 users in 1,000)
- You get swelling of the hands, feet, ankles, face, lips or throat which may cause difficulty in swallowing or breathing. You could also notice an itchy, lumpy rash (hives) or nettle rash (urticaria). This may mean you are having an allergic reaction to Cortimax.
- You pass black tarry stools or notice fresh or clotted blood in your stools (faeces). You may also notice dark bits that look like coffee grounds in your vomit. These could be signs of a stomach ulcer. Not known (frequency cannot be estimated from the available data)
- You get severe stomach pain which may reach through to your back. This could be a sign of pancreatitis. Serious effects: Tell a doctor straight away if you notice any of the following side effects: Steroids including Cortimax can cause serious mental health problems. These are common in both adults and children. They can affect about 5 in every 100 people taking medicines like Cortimax.
Serious effects: Tell a doctor straight away if you notice any of the following side effects.
Uncommon (affects 1 to 10 users in 1,000)
- Feeling depressed, including thinking about suicide.
Not known (frequency cannot be estimated from the available data)
- Feeling high (mania) or moods that go up and down.
- Feeling anxious, having problems sleeping, difficulty in thinking or being confused and losing your memory.
- Feeling, seeing or hearing things which do not exist. Having strange and frightening thoughts, changing how you act or having feelings of being alone.
- Pheochromocytoma crisis (symptoms can include an awareness of your heart beat, increase in heart rate (palpitations), excessive sweating, high blood pressure, severe headaches or have a tremor (feeling shaky)). Other serious side effects include: Not known (frequency cannot be estimated from the available data)
- A very sore throat. You may also have difficulty in swallowing and the inside of your mouth may have white areas on the surface.
- Headache, which is usually worse in the morning, on coughing or straining, and feeling sick (nausea). Also, fits, fainting, eyesight problems, painful eyes or confusion can occur.
- In infants with a low birth weight a heart muscle disease (hypertrophic cardiomyopathy) may occur. If you notice any of these problems talk to a doctor straight away. Other side effects: Please tell your doctor or pharmacist if any of the following side effects gets serious or lasts longer than a few days.
Uncommon (affects 1 to 10 users in 1,000)
- Stomach or bowel problems such as feeling full or bloated, indigestion, heartburn or stomach pain.
- Increase in appetite and weight gain including around your face. Or, you may lose weight or feel weak.
- Hair, including body or facial hair, grows more than normal.
- Increased thirst and needing to pass water more often than usual. These could be signs of diabetes. If you are already diabetic, your doctor may prescribe more of your diabetes medicine to balance the effects of deflazacort. You should discuss this with your doctor.
- Raised blood pressure and increased water retention.
- Tiredness, confusion, muscle weakness or muscle cramps. This may be due to low levels of potassium in your body.
- Mood changes, difficulty in sleeping.
- If you have had tuberculosis (TB) in the past it may return.
- Skin problems such as acne, appearance of stretch marks.
- You may get infections more easily than usual. Rare (affects 1 to 10 users in 10,000)
- Bleeding under the skin, redness.
- General muscle weakness or tiredness. Not known (frequency cannot be estimated from the available data)
- Bones and tendons may break or tear more easily than usual. Also tendons may get inflamed and become painful.
- Irregular periods in women or they may stop altogether.
- Becoming dependent on deflazacort (also called psychological dependence).
- If you have schizophrenia your symptoms may get worse.
- Fungal infection such as thrush.
- Eye disease that causes detachment of the retina and bulging eyes.
- Eye problems such as glaucoma and cataracts can happen if you take this medicine for a long time.
- Eye infections (viral) may spread or return if you have had them in the past.
- Blurred vision.
- Increase in the risk of clots forming in your blood.
- Blood problems such as leukocytosis.
- Wounds and cuts do not heal as quickly as usual.
- Noticeable blood vessels, thinning of the skin.
- Sudden or severe muscle weakness or tiredness following an operation.
Some of the side effects are more likely to happen if you are elderly. Children and teenagers taking this medicine may grow less than normal. (Not known: frequency cannot be estimated from the available data). If you think this is happening to a child, tell your doctor.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly: Website: www.zuventus.com and click the tab “Safety Reporting” located on the top of the home page.
By reporting side effects, you can help provide more information on the safety of this medicine.
You can also report the side effect with the help of your treating physician.
5. How to store Cortimax
Keep out of the sight and reach of children. Store in a dry place at a temperature
not exceeding 25°C.
Do not take this medicine after the expiry date, which is stated on the carton after “EXP”. The expiry date refers to the last day of that month.
Keep it in the pack in which it was given to you. Do not transfer your medicine to another container.
Do not dispose of medicines by flushing down a toilet or a sink or by throwing out with your normal household rubbish. This will help to protect the environment.
6. Further information
What Cortimax contains
10 Strips of 6 tablets in each strips
- Each tablet contains Cortimax 6mg/12mg/24mg/30mg of the active substance, Deflazacort.